Challenges within Life Science
The pursuit of new breakthroughs in pharmaceutical and medical technology is a daring endeavor that demands unwavering commitment and substantial resources. It is not only essential for these innovations to be effective and beneficial to patients, but they must also ensure patient safety during large-scale production.
The life science industry operates within a framework of stringent regulations and controls to ensure that standards and requirements are upheld. Regulatory authorities impose strict demands for documentation and traceability throughout the different stages of a product's life cycle.
The abundance of documentation and the necessity for control frequently result in cumbersome manual administration and inadequate internal processes without proper system support. Consequently, this leads to challenges in locating information, inadequately documented internal procedures, and insecure storage of vital data.
Centuri as solution
Centuri is an innovative and customizable solution that adheres to the highest industry standards. By choosing Centuri, you can eliminate a significant portion of the validation process that is typically required with in-house developed systems.
Centuri not only enables you to meet all regulatory requirements, such as controlled access, life cycle management, and traceability, but also empowers you to effortlessly find, organize, and maintain documents. It provides a comprehensive management system that effectively addresses risks, deviations, and corrective actions while ensuring competence and authorizations. With Centuri, you can streamline your processes and enhance overall efficiency in the most seamless way possible.
With over two decades of expertise in life science management systems, Centuri has been meticulously crafted to effortlessly integrate into smaller businesses while still possessing the robust capabilities to support global scalability.


















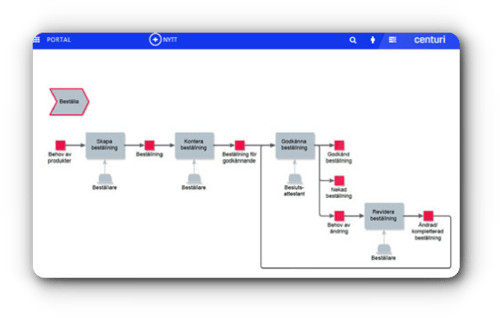
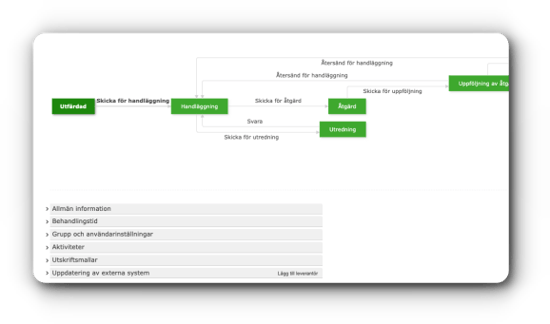
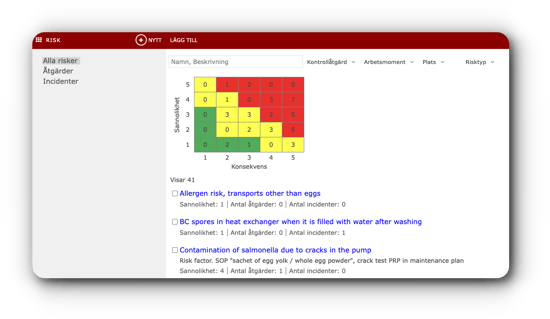 Centuri empowers you to securely oversee and control your instructions, routines, product documentation, and checklists.
Centuri empowers you to securely oversee and control your instructions, routines, product documentation, and checklists.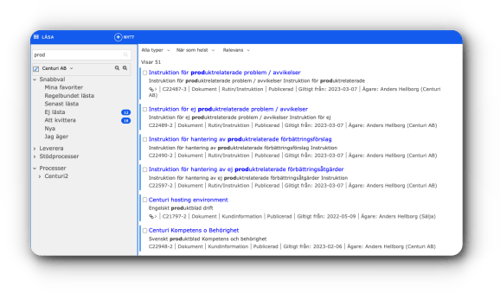 Empower your workforce to steer clear of needless work.
Empower your workforce to steer clear of needless work.